CWD Tuberculosis Found in Spongiform Disease Formerly Attributed to Prions: Its Implication towards Mad Cow Disease, Scrapie and Alzheimers
Lysenko AP PhD,Broxmeyer L MD, Vlasenko VV PhD, Krasochko PA PhD, Lemish AP and Krasnikova EL
Lysenko AP PhD1, Broxmeyer L MD2, Vlasenko VV PhD3, Krasochko PA PhD1, Lemish AP1 and Krasnikova EL1
1 Institute of an Experimental Veterinary Science, n. S.N. Wyshelesski, Belarus
2N.Y. Institute of Medical Research in Bayside, New York, USA
3Podolsk Center of Tuberculosis, Ukraine
- *Corresponding Author:
- Broxmeyer L
N.Y. Institute of Medical Research in Bayside,
New York, USA.
Tel: 011-718-229-3694
E-mail: drlawrencebroxmeyermd@alumni.usc.edu
Received date: April 05, 2017; Accepted date: April 27, 2017; Published date: April 29, 2017
Citation: Lysenko AP, Broxmeyer L MD, Vlasenko VV, et al. CWD Tuberculosis Found in Spongiform Disease Formerly Attributed to Prions: Its Implication towards Mad Cow Disease, Scrapie and Alzheimer’s J Mol Path Epidemol. 2017, 2:3.
Abstract
The TSE’S or transmissible spongiform encephalopathies, include bovine spongiform encephalopathy (also called BSE or “mad cow disease”), Creutzfeldt– Jakob disease (CJD) in humans, and “scrapie” in sheep or goats(caprine spongiform encephalopathy). They remain a mystery, their cause still hotly debated. Current madcowdiagnosisliessolely inthedetectionoflateappearing “prions”, anacronym for hypothesized, geneless, misfolded proteins, somehow claimed to cause the disease. Yet laboratory preparations of prions contain other things, which could include unidentified bacteria or viruses. And the only real evidence that prion originator Stanley Prusiner had in his original paper that the disease agent behind “Scrapie” in sheep and goats was devoid of DNA or RNA– was based upon the fact that he couldn’t find any. Furthermore, the rigors of prion purification alone, might, in and of themselves, have killed any causative microorganism and Heino Dringer, who did pioneer work on their nature, candidly predicts “it will turn out that the prion concept is wrong.” Roels and Walravens as well as Hartly traced Mad Cow to Mycobacterium bovis. Moreover, epidemiologic maps of the origins and peak incidence of Mad Cow in the UK, suggestively match those of England’s areas of highest bovine tuberculosis, the Southwest. The neurotaxic potential of bovine tuberculosis has for some time been well known. By 1911 Alois Alzheimer called attention to “a characteristic condition of the cortical tissue which Fischer referred to as ‘spongy cortical wasting” in Alzheimer’s disease (AD). But behind AD, Fischer suspected a microbe called Streptothrix which was constantly being mistaken and confused for tuberculosis. Our present investigation of the TSEs clearly shows cell-wall-deficient (CWD) tubercular mycobacteria present, verified by molecular analysis, ELISA, PCR and microscopy to cause spongiform encephalopathy
Keywords
Prions; Scrapie; The Spongiform Encephalopathies; Alzheimer ’s disease; The etiology of Alzheimer’s Disease; Mycobacterium tuberculosis Complex
Introduction
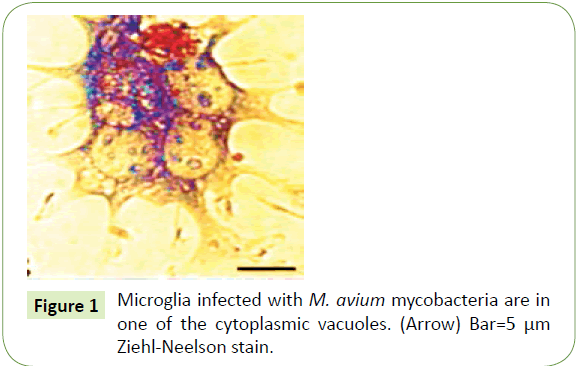
With the transmissible spongiform encephalopathies (TSE’s), including “scrapie” and “mad cow” in animals- mental and physical abilities deteriorate and myriad tiny holes appear in the brain’s cortex causing it to appear like a sponge (hence spongiform). Such "spongiform change" is caused by intracellular vacuoles in neurons and glia. Yet through eons of evolution and as a survival strategy it is through the formation of just such intracellular vacuoles that TB and the mycobacteria, unlike most pathogens, have survived, adopted, and multiplied either in the macrophages of the body or the neurons and glia of the central nervous system [1-3]. The very same intracellular vacuoles that eventually lead to spongelike changes in the brain form in microglia within a few hours of an infection with fowl tuberculosis (Mycobacterium avium) – which shifts its morphology from thick short mycobacteria to the longer filamentous forms so often documented in the spongiform encephalopathies [1]. And a similar scenario is operative for M. tuberculosis and other mycobacteria, all of which thrive in these vacuoles, possibly creating a reservoir in the brain through which additional microglia and neurons can be infected.
Today, the TSE’s are thought to be caused by “prions” characterized by neuronal loss, gliosis, spongiform change and intracellular vacuoles in neurons and glia- yet these very same changes are instigated by tuberculosis and its related mycobacteria. Microglia internalizes virulent M. tuberculosis more rapidly and efficiently than less virulent strains. Upon internalization, tubercle bacilli are found in sparse, but densely packed vacuoles (Figure 1).
Clinically what is seen in all the spongiform encephalopathies is a rapidly progressive dementia, and their most common clinical presentation in humans is CJD or Creutzfeldt–Jakob disease as well as in certain cases of Alzheimer’s disease. In the opinion of experts, ample justification exists for considering a similar pathogenesis for Alzheimer’s, Creutzfeldt–Jakob, scrapie and the other spongiform encephalopathies such as Mad Cow disease. In fact, Creutzfeldt–Jakob and Alzheimer’s often coexist and at this point are thought to differ merely by time-dependent physical changes. One study linked up to 13% of all Alzheimer’s victims as really having Creutzfeldt-Jakob disease [4].
In 1909, Sir Stewart Stockman, Chief Veterinary Officer of the Royal College of Veterinary Surgeons, while investigating a disease of sheep in Scotland known locally as “scrapy” or “scrapie”, found acid-fast bacilli characteristic of fowl or cattle tuberculosis in lesions of animals dying from scrapie [5]. Stockman will later be mentioned as one of the historical figures in the discovery of prion disease, but the fact that he found acid-fast bacilli in scrapie, a premier finding for tuberculosis, would not.
By 1911 Alois Alzheimer noted in some Alzheimer’s cases “a characteristic condition of the cortical tissue which Fischer has referred to as ‘spongy cortical wasting.” Alzheimer was referring to the finding of his great rival, neuropathologist Oskar Fischer of Prague. Fischer named and was one of the first to describe such spongiform cortical atrophy [6]. But Fischer was investigating such Alzheimer’s brain changes in the context of being from a microbe called Streptothrix, which was constantly being mistaken and confused for tuberculosis. And just ten years before Oskar Fischer found Streptothrix-like forms in Alzheimer’s cerebral plaque, BabèÃÆââ¬Â¦Ãâà ¸ and immunologist Levaditi reported that once inoculated with tubercle bacilli beneath the dura mater of their brains, rabbits not only developed branched filamentous forms similar to Streptothrix but rosettes that were identical to the "drüsen" that Oskar Fischer repeatedly reported in Alzheimer’s plaque [7].
By 1936, George W. Dunkin reviewed Stockman’s findings, confirming acid-fast organisms in the “scrapie” of sheep and goats. But unlike the bovine condition known as paratuberculosis (a subspecies of fowl tuberculosis or Mycobacterium avium found mainly in cattle), the acid-fast forms of TB confirmed by Dunkin in “scrapie” were longer and more slender then those encountered in paratubercular Johne’s disease in cattle.
Not only is bovine tuberculosis (Mycobacterium bovis or M. bovis) capable of infecting all animals and men subject to the spongiform encephalopathies but by 1868 Jean-Baptiste Auguste Chauveau in France and Edwin Klebs in Prague proposed that eating infected meat could transmit bovine tuberculosis [9,10]. Subsequently Andreas Gerlach’s experiments confirmed that either milk or flesh from tubercular cows could transmit tuberculosis to other animals to which they were fed [11]. Schliesser also saw meat from tuberculous animals as constituting a significant risk to humans if available for consumption [12]. With regard to bovine tuberculosis, anyone handling or consuming meat from an infected animal is at risk of contracting this disease. By the turn of the 20th century 25% of the many US deaths from TB in adults were caused by M. bovis.
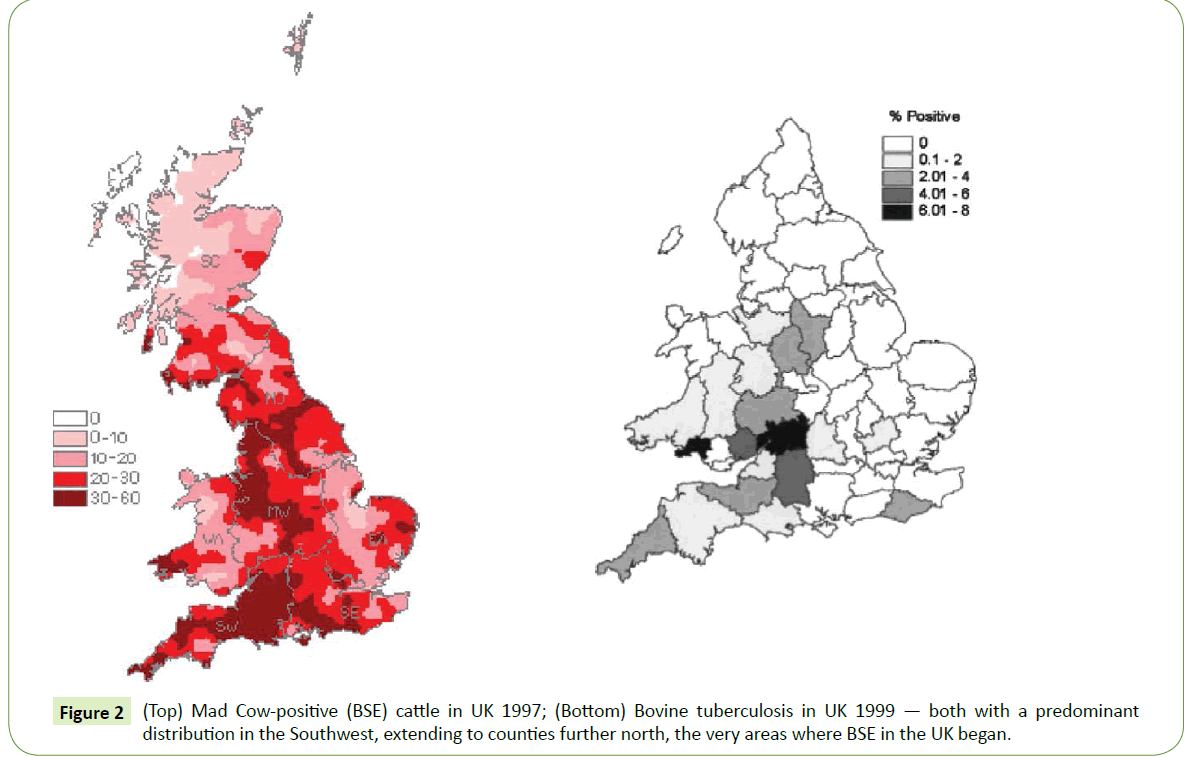
While various theories continued to swirl around the cause of TSE’s, the best epidemiologic maps of the peak incidence and prevalence of BSE or mad cow disease, done in the UK, it turns out, suggestively matched those of the highest prevalence of England’s bovine tuberculosis in cattle, with a predominant distribution in the Southwest (Figure 2) extending to counties further north- the very area where BSE in the UK began [13,14].
The prion hypothesis
The TSEs or transmissible spongiform encephalopathies, are currently alleged to be caused by “prions.” But the theory surrounding neurologist Stanley B. Prusiner’s “prion”, a word which he himself coined for gene-less proteins that were infectious, was under a rightful cloud of suspicion from its onset [15].
All living organisms, including germs, use their DNA and RNA to make copies of themselves and amplify themselves in the brains of those they infect. But prions supposedly did not have DNA or RNA. Therefore, Prusiner’s theory was felt to be heretic, unorthodox, and contrary to accepted belief. And the only real evidence that he had in his original paper was that the disease agent behind “scrapie” was devoid of DNA or RNA– was because he couldn’t find any. Not only is scrapie difficult to diagnose but it is always fatal, a degenerative disease that affects the nervous systems of sheep and goats. The name “scrapie” is derived from one of the clinical signs of the condition, wherein affected animals will compulsively scrape off their fleeces against rocks, trees, or fences. The disease apparently causes an itching sensation in the animals. Scrapie is the most common reportable disease in goats and sheep in the United States today.
Previous studies in which he had been involved in suggested to Prusiner that there may be similarities between the agents causing scrapie encephalopathy in goats and Creutzfeldt-Jakob disease (CJD), a rare but fatal disease in humans [16]. Yet Prusiner’s initial 1980 scrapie paper, done with Hadlow, Kennedy, and Race mentions absolutely nothing about prions, referring instead to “unconventional viruses”, “the CJD virus” and “the scrapie virus”.
All things considered, Prusiner’s idea wasn’t new. The thoughts of biologists in the 1930s who incorrectly said that viruses were only proteins and that ‘slow viruses’ might be gene-less had been proposed by and discarded in Britain by Griffith as early as 1967 [17]. To be certain, British mathematician J.S. Griffith was first to propose not only a self-replicating protein theory, but that proteins could be chemically identical but simply folded differently.
The finding that prions were proteins normally found in the body, including the brain of healthy controls, seemed to contradict the best evidence that they were infectious. The theory, like all questionable science, survived by finding a difference: prions from healthy animals were quickly claimed to be “cellular” protein; those from scrapie were “scrapie” protein. Scrapie protein aggregated into rods while cellular protein did not. Another socalled “critical clue” [18]: scrapie protein survived proteinase, while “cellular” did not. This still did not mean however that some virus or bacteria or mycobacteria did not cause the change being attributed to prions to begin with. Cow tuberculosis (Mycobacterium bovis), for example, both by virtue of its cellwall- deficient, viral-like forms, and that it shares methyllysines with other mycobacteria is also protease-resistant [19]. Did this mean that tuberculosis was also geneless?
The amyloid proteins in Alzheimer’s, which as early as his 1982 Scrapie paper Prusiner was suggesting be linked to prions, were also known at that time to be protease resistant, but not from prions [20].
By all logical estimates, the death-knell to the prion hypothesis should have occurred with Lasmézas’s 1997 interspecies transmission of mad cow in which more than half of injected mice had no detectable prions [21]. If this was not enough, then there was Baker and Manuelidis’s 2002 study on infectious neurons (microglia) with low prion levels in otherwise highly infectious material, which only served to support the concept that pathologic prions were the result of infection rather than being the actual infectious agent [22].
When Yale’s Laura Manuelidis and Sotirios Botsios revisited this subject in 2016 they outright stated that CJD and Scrapie require agent-associated nucleic acids for infection, therefore turning the entire original prion hypothesis on its head [23]. Manuelidis called for a re-opening of the entire prion concept to find out just which genetic material was involved, and its origin. To Manuelidis this was likely to be a virus, although she admitted the fundamental mystery remained. In fact, to many dissenters, some other, not as yet identified pathogen such as a virus, bacteria, or mycobacteria caused “prions” to misfold — thus damaging the brain.
Cell-Wall-Deficient Mycobacteria
Long before the prion theory was proposed, scientists searched unsuccessfully for the virus or viruses that might cause TSE, including Dr. Stanley Prusiner. And even today, some speak in terms of the TSEs (transmissible spongiform encephalopathies) being caused by actively replicating infectious particles of viralsize and density. But “viral-sized” does not necessarily mean virus.
Tuberculosis is an infectious disease that is found throughout the world and which can have a dramatic impact on the economy and on human and animal health. In humans, the principal cause of infection is Mycobacterium tuberculosis, but the animal-specific pathogen, M. bovis, is arguably the most important zoonotic agent in human history [24].
Tuberculosis due to M. bovis has a worldwide distribution, and represents one of the most important public and veterinary health problems [25]. It is of greatest concern in cattle even though it can occur in humans and many domesticated and wild animal species including goats, swine and sheep. M. bovis is an important factor in human tuberculosis [26]. Furthermore, in at least one form of tuberculosis in sheep and goats ÃÆâÃâââ¬Ãâââ¬Â¢ paratuberculosis, the wool or hair is often damaged and easily shed, not unlike in scrapie. Signs are rarely evident until two or more years after the initial infection, which usually occurs shortly after birth. And once symptoms appear, paratuberculosis is progressive and affected animals eventually die.
Of all the bacteria, the preferred and predominant form of the mycobacteria is their cell-wall-deficient “L” forms which are crucial to their survival in the body. And it is here that diagnosis becomes most problematic [27]. Mycobacteria are difficult to detect, especially in their viral-like, obscure state. Similar to prions, mycobacterial L-forms escape destruction by the body’s immune system, and are seemingly indestructible. Tubercular L-forms, no matter their exact mycobacterial species, are remarkably different from L-forms of other bacteria in their resistance to chemical and physical agents [28,29]. And the smallest and most resistant to such environmental stresses are their filterable tiny DNA containing L-granules – although other polymorphic viral-like and staph-like coccoid forms also form [30]. Yet genetic analysis by16S rRNA 16S ribosomal RNA confirms them as tubercular. Despite what is referred to as prions “heterogeneity with respect to size” ÃÆâÃâââ¬Ãâââ¬Â¢ this is also a well-known characteristic of polymorphic CWD mycobacteria, with their diminutive L-granules mimicking resistant “small proteinaceous particles” in every way. So the fact that CWD tuberculosis is protease resistant or “resistant to inactivation by most procedures that modify nucleic acids” comes as no surprise without the need to label them “proteaseresistant proteins.”
It is in the relatively dormant state of cell-wall-deficiency that the mycobacteria, not unlike prions, develop enhanced resistance to harmful factors and a long life- a survival strategy that has allowed the mycobacteria to become one of the most successful pathogens on the planet. But the downside is that such dormant cell-wall-deficient or “L-forms” also become among the most difficult to cultivate and identify, especially in their early noncultivable or so-called “invisible” stage [31]. Therefore to find them in the living organism (in vivo) takes mandatory novel strategies including special growth techniques to enrich and revive them to an actively growing, colony-forming state. To Zhang this meant the resuscitation of dormant Mycobacterium tuberculosis by phospholipids or specific tubercular protein peptides [32]. However, the same CWD growth stimulation can occur without the use of mycobacterial elements, such as the use of growth stimulants which create nutrient starvation or hypoxic conditions for M. tuberculosis in vitro [33]. With either methods, CWD mycobacteria – otherwise non-cultivable, will grow, often transitioning from ovoid or egg-shaped to rod shaped cells. In fact, the recovery of the ability of mycobacteria to cultivate has traditionally been associated with the formation of the same rod-shaped, filamentous and granular forms that make CWD tubercular forms indistinguishable from prions [34]. We used a mycobacterial Cell-Wall-Deficient growth stimulant without any mycobacterial components.
Amyloid: the common denominator in all Spongiform Encephalopathies
“It is an astounding finding, because we never would have dreamed that amyloid and prions are the same”, proclaimed Stanley Prusiner [35]. But previous to this and throughout the history of medicine amyloid was usually considered the deposition that took place due in the course of chronic inflammatory disease (with genes), mainly tuberculosis- its usual precipitating cause.
Hass’s study at Cornell proved a direct correlation between amyloid deposition and the mycobacteria by injecting M. bovis into rabbits and following M. tuberculosis in humans. He concluded that the only infectious disease which served as an apparent cause of amyloidosis was tuberculosis [36]. All of the 21 human subjects with amyloid in Hass’s investigation had chronic pulmonary tuberculosis.
In addition, in a 50-year study based upon autopsy, Schwartz saw amyloidosis, primary and secondary, in the brain and elsewhere as a by-product of underlying infectious tuberculosis, either reactivating itself or being reactivated by a host of traumatic, chemical, biologic or physical insults [37]. Microscopically, in the brain, Schwartz found plaques and amyloid degeneration of nerve fibrils. When Schwartz injected 22 guinea pigs with M. tuberculosis, all but four came down with amyloidosis. His uninfected controls, with the exception of one, showed no amyloid. He thereby confirmed Hass, whose large series of rabbits showed that three out of four inoculated with bovine tuberculosis had amyloid disease within 15 months [36]. Hass’s amyloid uniformly showed a principal protein fraction.
The amyloid issue surfaced again when in 1978, Researcher Pat Merz, in breakthrough work, identified tiny fibrils in the brains of scrapie infected mice not present in well controls. Prion purists refused to admit that their prion rods were related to Merz’s find, citing her entities as longer fibrils and claiming that Merz stated plainly that her scrapie-associated fibrils (or SAF) were not amyloid and therefore could not be prion rods – the term Prusiner used for amyloid fibrils. Actually Merz said that her scrapie associated filaments were amyloid-like on more than one occasion and other workers in the field suggested that the two entities in Merz and Prusiner’s papers were identical [15]. Delgado saw such fibrils in either case as typical features of amyloid [38].
Meanwhile, by 1994, de Beer, studying the relationship between a major rise of serum amyloid and having tuberculosis, saw a rapid descent in amyloid in patients treated with anti-tubercular drugs [39]. As an offshoot of de Beer’s work, Tomiyama dissolved β-amyloid plaque with rifampin, a first line drug in the treatment of TB, and one of the few agents, to this day, that is able to dissolve amyloid plaque [40].
Whether in humans or animals, it is impossible to address tuberculosis without speaking of its cell-wall-deficient or “L-forms” which immediately start to form in the body’s macrophage and microglial defenders once under attack and continue to propagate thereafter [41-43].
The purpose of this work was therefore to establish whether central nervous system CWD tubercular infection could result and account for in the clinical and histopathological changes that we associate with transmissible spongiform encephalopathies (TSEs) .
Materials and methods
Transmissible Spongiform Encephalopathy
Fragments from the brains of two goats with advanced clinical damage to the central nervous system and clinical signs of scrapie were fixed in 15% formalin (1 month). Laboratory diagnostics already performed had ruled-out rabies or listeriosis. Brain fragments, after being washed with formalin in sterile water were homogenized and incubated on a mycobacterial growth stimulant (1:4) containing 0.1% chlorhexidine for 48 hours, allowing for the growth and eventual isolation of cell-wall-deficient (CWD) tubercular/mycobacterial forms [44-46]. This was then planted in test tubes with a solid mycobacterial nutrient media containing agar, asparagine, peptone, glutamate, and the elements K, Mg, Zn, Mn, Fe and S and P incubated at 37°C for an additional 10 days. A repeat observation and mycobacterial probe was conducted after 5 years of preservation in formalin in Brain 1 and after 3 years in the case of Brain 2.
Microscopy
Histological sections of the cortex and other sites of the brain were prepared by standard technique and stained with Hematoxylin- Eosin stain, Ziehl-Neelsen, and modified immunoenzyme technique (MIET) [45,47]. MIET staining procedure included fixing at 65°C for 2 hours followed by the suppression of endogenous peroxidase with 3% H2O2. Staining by Kinyoun carbol-fuchsin was done for 5 minutes, followed by decolorization with 3% acid alcohol for 70 seconds, counterstained by 0.3% methylene blue for 2 minutes. Further conjugates of affinity purified antibody to M. bovis were drawn and then placed in Phosphate-buffered saline (PBS) with 0.02% Tween 20 for 1½ hours followed by emersion into a substrate solution consisting of 10 mg. 3,3'-diaminobenzidine (Fluka Sigma-Aldrich) in 2 ml of DMSO and 10 ml of deionized water with 10 μl of 33% H2O2 for twenty minutes.
At each stage, smears were washed with deionized water with 0.02% of Tween 20. MIET (modified immunoenzyme technique) stained slides showed either classical acid-fast (AF) tubercular bacilli stained in red, or non-acid-fast (NAF) cells or antigens to mycobacteria in brown. Other microflora and tissue cells were stained blue. Results of microscopy were recorded using an Olympus 51BX microscope.
Cell-Wall-Deficient (CWD)
Tubercular reference standards used included isolates from the 1stInternational standard PPD for Mycobacterium bovis, isolates from the blood of a TB patient, isolates from the lymph node of a tubercular cow, isolates from embryonic (fetal) bovine heart tissue, and CWD forms of M. bovis BCG and M. bovis Vallée. That all CWD isolates were from the genus Mycobacteria was confirmed both by the existence of common antigens with reference strains of M. bovis, M. avium-paratuberculosis complex and M. tuberculosis as well as PCRs with primers for 16S rRNA, MPB70 – a major protein antigen of Mycobacterium bovis, and primers for M. avium-paratuberculosis complex.
SDS-PAGE electrophoresis and Western blotting
Mycobacterial CWD isolates were washed three times with a 0.5% phenol solution suspended in stock buffer and warmed for 5 minutes at 99°C. Lysates of isolates were separated by SDS-PAGE electrophoresis in 15% of PAGE (Laemmli, 1970).
Polymerase Chain Reaction (PCR)
The identification of isolates was carried out by 16S rRNA mycobacterial primers, MPB70 complex tuberculosis-bovis primers and primers for Mycobacterium avium-paratuberculosis complex (Pramytech). Suspensions of isolates in lysing buffer were warmed for 5 minutes at 95°C. The DNA was isolated on columns with a sorbent (IBOH NANB) and amplification was carried out on a C1000 TouchTM Thermal Cycler (BioRad) according to standard protocols. An electrophoresis of amplificates was then carried out in 2% agarose gel and results obtained from a Molecular Imager®GelDocTM XR+system. (BioRad).
ELISA
ELISA of the sonicate isolates from both brains were done with the rabbit Antisera to sonicates of M. tuberculosis H37Rv, M. bovis Vallee and M. avium 1603.
Results
Spongiform changes found in tubercular cortex
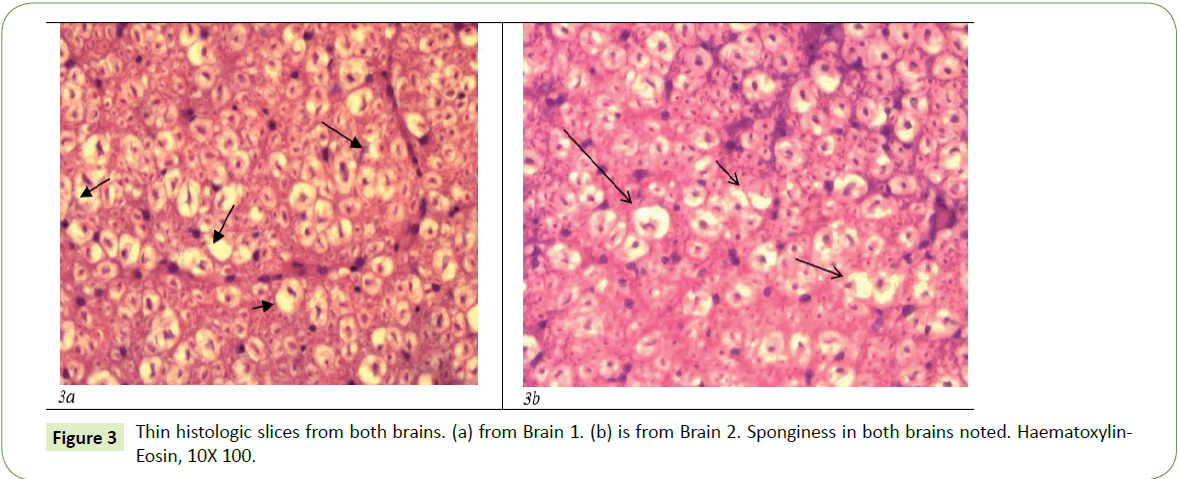
Multiple brain thin cuts were performed at the level of the cerebral cortex and the pons in two tubercular goats which before dying demonstrated the clinical signs of advanced transmissible spongiform encephalopathy (scrapie). Spongiform changes caused by intracellular vacuoles, accompanied by thrombotic vessels, proliferation of cerebral vessels, perivascular cuffing, and lymphocyte infiltration were all in evidence. In addition to porous spongiform changes there was also a proliferation of heavily vacuolated glia (Figure 3).
Cell-Wall-Deficient tubercular forms
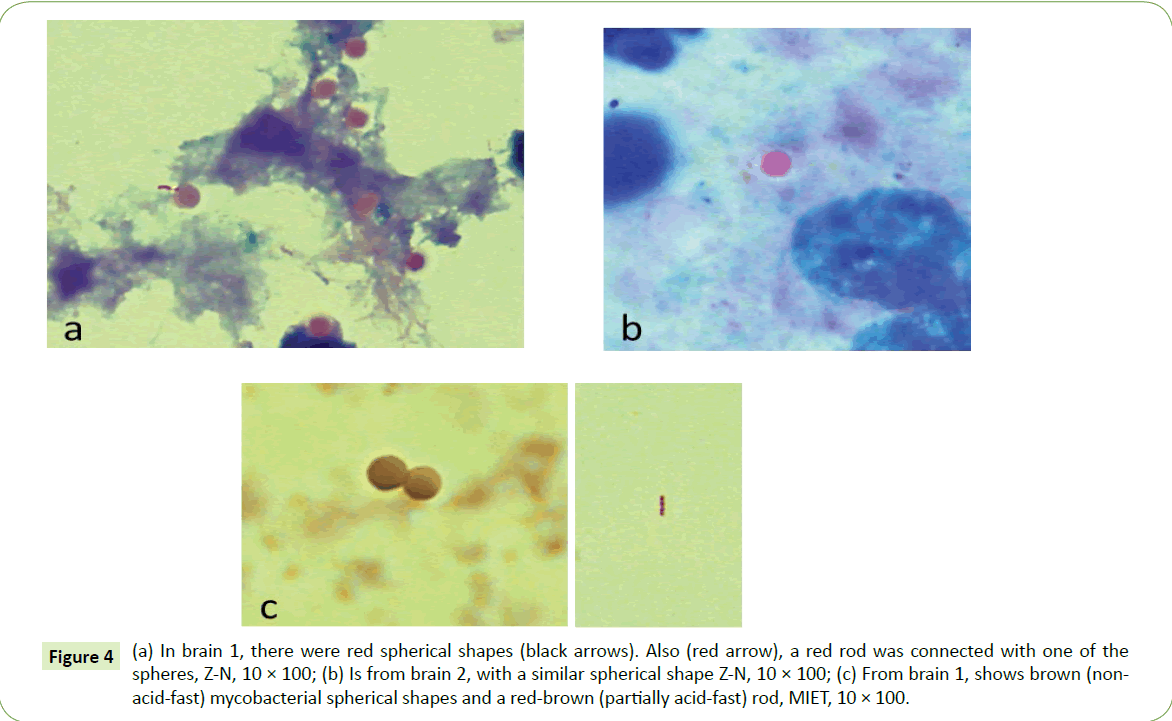
At microscopy, some of the smears of a brain stained with Ziehl–Neelsen (ZN) stain yielded various cell-wall-deficient mycobacterial forms. Among these were partially acid-fast spherical shapes (Figure 4a-4c) which were greater in number in Brain 1, and rarer in Brain 2. In one such case, a partially acid-fast sphere (spheroplast) is actually seen connected to a partially acid fast (red color) rod (pink arrow in Figure 4a). MIET stained these spheroplasts brown, indicating non-acid-fast (NAF) mycobacterial forms (Figure 4c). Also seen in Figure 4c is one of the occasional acid-fast rods seen (Figure 4c).
Figure 4: Thin(a) In brain 1, there were red spherical shapes (black arrows). Also (red arrow), a red rod was connected with one of the spheres, Z-N, 10 × 100; (b) Is from brain 2, with a similar spherical shape Z-N, 10 × 100; (c) From brain 1, shows brown (nonacid- fast) mycobacterial spherical shapes and a red-brown (partially acid-fast) rod, MIET, 10 × 100.
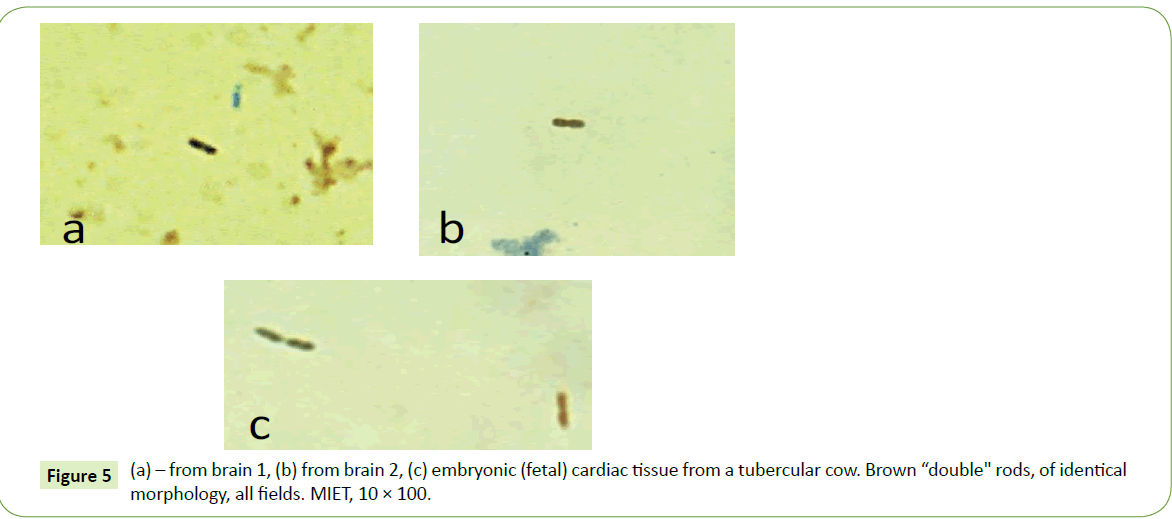
MIET staining of brain specimens also revealed brown rods (Figure 5). Such rods were more prevalent in Brain 2. The rods found in both brains had almost identical morphology (Figure 5); as well as having a double rod configuration of similar morphology. Double rods with similar morphology were also found in tubercular embryonic (fetal) cardiac smears obtained from a tubercular cow (Figure 5c).
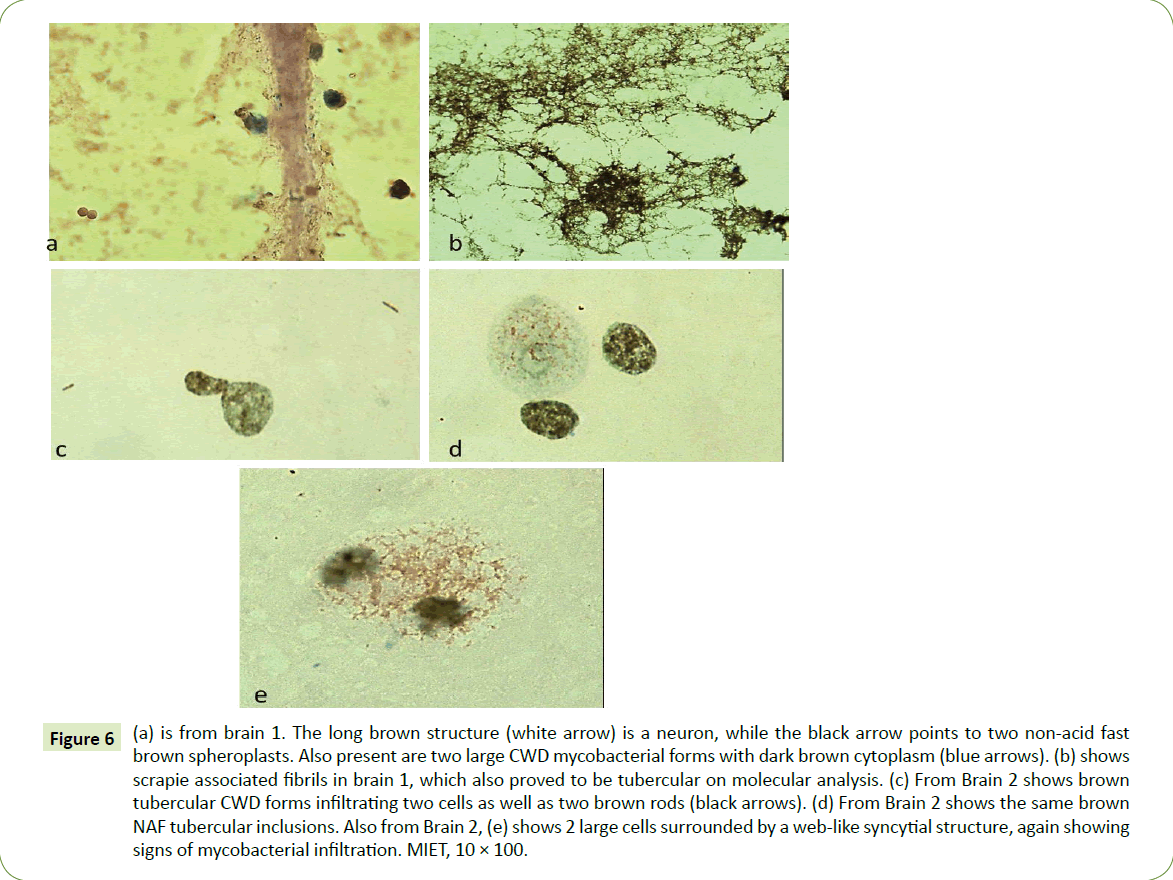
Antibodies to M. bovis reacted with autopsied tissue from both animals with advanced scrapie, making possible the visualization of other commonly encountered tubercular cell-wall-deficient (CWD) forms and mycobacterial antigens. In Figure 6a, the white arrow points to a neuron while various CWD non-acid fast mycobacterial inclusions (brown) in the cytoplasm can be seen in Figures 6a-6e. Such brown inclusions in the cytoplasm (Figures 6a-6e) are often surrounded with a brown web-like syncytial network (6e). Figure 6b shows brown fibrils similar to scrapieassociated- fibrils (SAF).
Figure 6: (a) is from brain 1. The long brown structure (white arrow) is a neuron, while the black arrow points to two non-acid fast brown spheroplasts. Also present are two large CWD mycobacterial forms with dark brown cytoplasm (blue arrows). (b) shows scrapie associated fibrils in brain 1, which also proved to be tubercular on molecular analysis. (c) From Brain 2 shows brown tubercular CWD forms infiltrating two cells as well as two brown rods (black arrows). (d) From Brain 2 shows the same brown NAF tubercular inclusions. Also from Brain 2, (e) shows 2 large cells surrounded by a web-like syncytial structure, again showing signs of mycobacterial infiltration. MIET, 10 × 100.
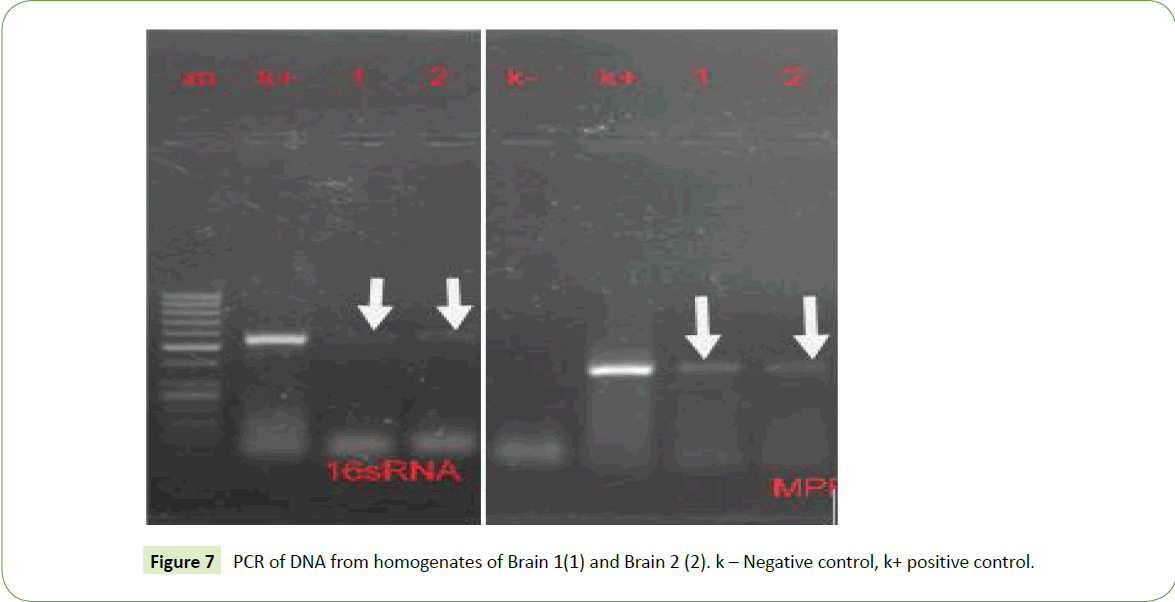
Fragments of brain tissue were washed in sterile water and homogenized. Upon PCR, primers for both 16sRNA and bovine tubercular MPB70 gave positive reactions (Figures 7).
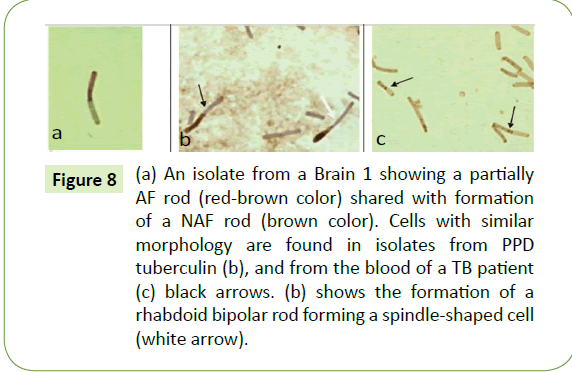
The rods of bovine tuberculosis are often spindle-shaped. After repeated subcultures, the color of the cells maintained a green shade that is connected with the weak synthesis of tubercular antigens. Isolates from both scrapie brains were polymorphic, yet shared common forms between themselves and CWD tubercular isolates from other sources. In smears, it was not unusual to see CWD mycobacterial forms in the same field that were long and short bipolar rods, thin granular rods, thick "empty" rods, and spindle-shaped forms (Figure 8). These same forms were found also in CWD isolates from tuberculin PPD [44], as well as from humans and cows sick with tuberculosis [44]. Very characteristically, as seen in Figure 9, are the forms of two rods seemingly bound together, with a septum in between – a configuration found in isolates from our brain segments, the tuberculins, and the blood of a tubercular patient, possibly representing a process of division. Such tubercular septation can create two daughter cells [48,49]. Again, the rods of the bovine tubercular bacillus are often spindle-shaped (Figure 8).
Figure 8: (a) An isolate from a Brain 1 showing a partially AF rod (red-brown color) shared with formation of a NAF rod (brown color). Cells with similar morphology are found in isolates from PPD tuberculin (b), and from the blood of a TB patient (c) black arrows. (b) shows the formation of a rhabdoid bipolar rod forming a spindle-shaped cell (white arrow).
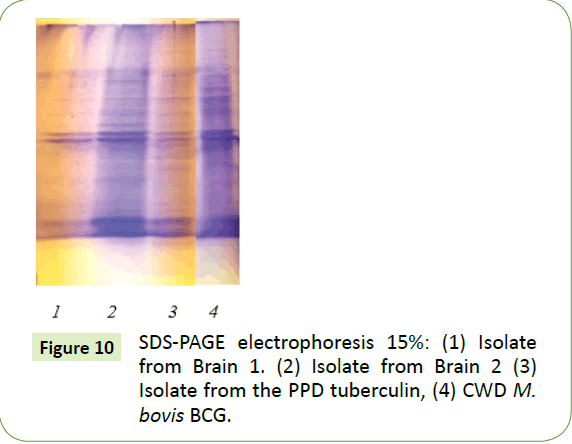
With SDS-PAGE electrophoresis, both brain isolates had identical polypeptide profiles (Figure 10, lane 1 and 2) which were, in turn, almost identical to both the profile for PPD tuberculin PPD (Lane 3) and that of a CWD M. bovis isolate (Lane 4).
In addition, Western blot revealed three amino acid (40-70 kDa) fractions from brain isolates reacted with antiserum to M. bovis, as well as similar fractions of isolates from standard PPD tuberculin of M. bovis (Figure 9). Similar triple fractions were also found in a typical MTB sonicate.
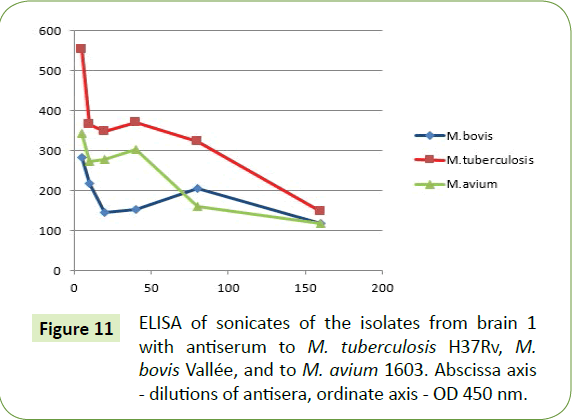
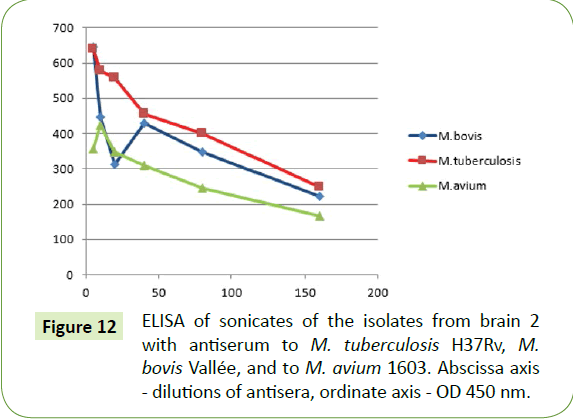
Upon ELISA, isolates from both brains reacted to the antiserums of M. tuberculosis, M. bovis and to a lesser extent M. avium (Figures 11 and 12).
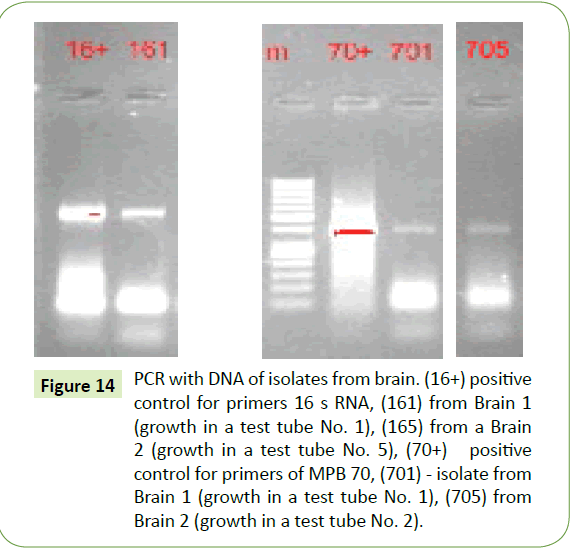
The subsequent formalin-resistance of the CWD tubercular forms taken from our brain specimens, even after 3 to 5 year exposure to 10% formalin were of particular interest. Brain samples (3-4 Gms) taken from formalin solutions under sterile conditions were placed in 100 ml of sterile water. These were then homogenized in 100 ml of sterile water. After 12 hours these were again homogenized with 10 ml of mycobacterial growth stimulant, incubated for 48 h at 37°C, and a 300 μl aliquot of this was then then planted in test tubes containing slanted semisolid mycobacterial growth medium. These test tubes were incubated at 37°C and after two days, smears from both specimens began to yield growth but visible colonies were not present. But under blind subculture at 24 hour intervals both series (Brain 1 and Brain 2) yielded small, transparent, merging colonies, the smears of which showed the same polymorphic rhabdoid spindle-shaped forms (Figure 13a) designating bovine tuberculosis as originally seen. And despite the large time interval of 3 to 5 years separating these assays from those of the original, they still reacted with the antibody of M. bovis, and the same mycobacterial PCR primers (Figure 14) and gave the characteristic brown-color for non-acidfast bacilli with MIET stain.
Figure 14: PCR with DNA of isolates from brain. (16+) positive control for primers 16 s RNA, (161) from Brain 1 (growth in a test tube No. 1), (165) from a Brain 2 (growth in a test tube No. 5), (70+) positive control for primers of MPB 70, (701) - isolate from Brain 1 (growth in a test tube No. 1), (705) from Brain 2 (growth in a test tube No. 2).
Discussion
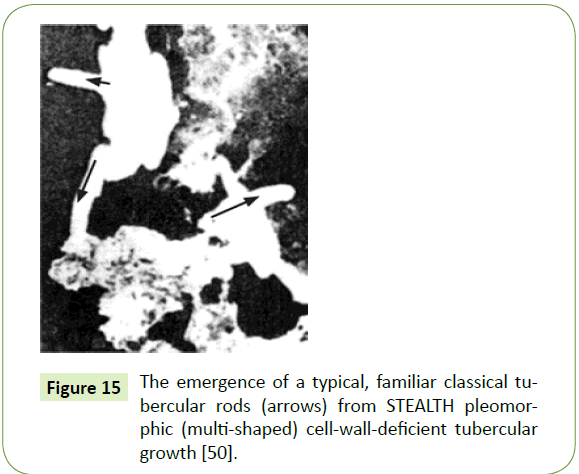
Spongiform and other pathological changes similar to BSE (Mad Cow Disease) were found in goats with the clinical symptoms of scrapie, who fell ill with signs of a central nervous system lesion in their brain. Yet in assays of both experimental animal’s brains, partially acid-fast tubercular cell-wall-deficient (CWD) spheroplasts, rods, and other mycobacterial CWD forms ÃÆâÃâââ¬Ãâââ¬Â¢ along with the existence of common tubercular antigens were found (Figure 15) [50]. Other known infectious diseases yielding similar brain lesions were excluded.
Besides this, net-like structures reacted with the antibody to M. bovis which were either the result of tubercular antigens or actual CWD tubercular forms developed a net-like syncytial structure similar to scrapie associated fibrils (SAF). Yet on the grounds of morphology, partial acid-fastness, the existence of antigens in common with MTB, and PCR/ELISA results, it was obvious that again, we were dealing with cell-wall-deficient and classical tubercular forms. Furthermore, our results simply confirmed studies done at the turn of the 20th Century, first by Stockman and later by Dunkin that spongiform changes in the brains of animals with scrapie were caused by acid-fast or partially acidfast tubercular bacilli [5,8].
Natural transmission of scrapie in the field seems to occur via the alimentary tract in the majority of cases, and scrapie-free sheep flocks can become infected on pastures where outbreaks of scrapie has been observed before. These findings point to a sustained contagion in the environment, notably in the soil [51]. Detectable “infectious” prions (PrPSC) have been reported in the feces of sheep both in the terminal and the early preclinical stages of the disease, suggesting the prions were likely to be shed into the environment throughout the course of the disease. But there is the precise same ability for transmission with tuberculosis and the mycobacteria through the alimentary tract as well as the sustained contagiousness of the Mycobacterium tuberculosis complex and related mycobacteria in the soil [52].
The length of time it takes for sheep or goats to develop scrapie is usually 18 to 24 months, a slow course which is not unusual in the progression of CWD tuberculosis. By 1996, Insanov warned that an infection with cell-wall-deficient forms of TB in children not only made standard treatment less effective but created a disease with a gradual, insidious onset and a slow accumulation of cerebral damage [53]. This made CWD tuberculosis more difficult to diagnose, because its slow burn into nervous systems, allowed months to years before its full spectrum of damage became obvious. Yet in this same study Insanov also showed that cell-wall-deficient forms in tuberculosis meningitis accounted for an incredible 87.6% of the tuberculosis found in children with TB meningitis and 87.3% of cases of TB meningitis in adults.
The original cause of cerebral tuberculosis, as well as other forms of the disease, can be as a result of exogenous infection or because of the activation of a latent tuberculous infection that crossed the placenta. We found evidence for this placental cross when the same tubercular rhabdoid, rod-like forms in our scrapie victims also appeared in the embryonic fetal tissue from a tubercular pregnant cow (Figure 5). In addition the lack of characteristic lung and lymph node involvement in our subject animals pointed towards a more systemic trans-placental infection.
Once a pathogen like tuberculosis has its cell wall disrupted, it become plastic, having the ability to assume many forms. Some are so tiny that they pass through a 22-μm filter, the so-called “viral stage” of a bacteria or mycobacteria. It is known that CWD TB can form various forms, including those with a size less than 0.22 μm (millimicrons) [33,54]. Because of this Cell-Wall-Deficient forms of MTB can penetrate not only the placenta but the bloodbrain barrier, and get into the brain. Calmette noted that after such infection with cell-wall-deficient tuberculosis, quick fetal death could occur. But even if the child were born alive, with no apparent ill-effects [55], their disease almost invisible – such stealth CWD forms of their dormant tuberculosis could spring back at any time to the diseases virulent forms.
Coming as no surprise, a similar and parallel failure to detect “prions” in the uterine and gestational tissues from a gravida with Creutzfeld-Jakob Disease has been reported [56].
Brieger, working at Cambridge University, demonstrated mainly pleomorphic stealth growth when he inoculated tuberculosis directly into the amniotic sac of lab animals [57]. The outer layer of the amniotic sac is part of the placenta. Rapidly, cell-walldeficient granules formed that did not stain with traditional acidfast stain. Within three days, other cell-wall-deficient forms such as the long branching fungal filaments of TB appeared ÃÆâÃâââ¬Ãâââ¬Â¢ easily mistaken for so-called “Scrapie Associated Fibrils” (SAF). Brieger repeated the study using fowl tuberculosis in chicken embryos. His results were similar [58]. Cell-wall-deficient forms appeared, again not identifiable by traditional stains or cultures.
Tubercular cell-wall-deficient forms, such as we isolated in scrapie are easily as resistant and long lasting as prions. Yet their proper isolation requires special mycobacterial growth stimulants and nutrient mediums [44-46]. The mechanism for the viability of tubercular CWD forms, even in the face of otherwise lethal thermal and chemical assault is a matter of active research, but just how this happens is still as yet undetermined. Our brain samples, which yielded such forms easily, withstood 3 to 5 years of storage in 10% formalin, comparable to the vaunted viability of prions. The formalin and high heat issues are no small part in the history of prions. Prusiner cited his predecessor, UK veterinarian Lain Pattison throughout his works saying at one point that Pattison was one of the first to suggest that the “scrapie agent” might be a basic protein “based on the results of his attempts to purify the infectious pathogen” [59]. Actually during the 1960s Pattison’s, even before mathematician Griffith, was astonished by the scrapie agents surprisingly high resistance to formalin as well as high heat [60] – properties which he and his collaborator Katherine Jones were certain that were closely similar to those of ‘encephalitogenic factor’, which Pattison believed to be ‘a basic protein’ [61]. But in reality, studies performed since Pattison suggest that formalin also does not effectively inactivate Mycobacterium tuberculosis or related mycobacteria in formalinfixed tissue [62,63] – certainly reflected by the fact that our brain samples continued to yield CWD tubercular forms – easily withstanding 3 to 5 years of storage in 10% formalin.
As life expectancy increases, with waning immunity, so too does the probability of acquiring an active TB infection of the CNS and elsewhere. Therefore Alzheimer’s most comprehensive 1911 paper was entitled: “On certain peculiar diseases of old age” [64]. Although Alzheimer’s disease, like Parkinson’s disease, is characterized by extracellular Aß plaque formation and intraneuronal tau protein inclusions – there is significant hesitancy to use the term “prion” to describe their occurrence. This is primarily because in contrast to CJD and Kuru, the transmission of Alzheimer’s and for that matter Parkinson’s has not been demonstrated between individuals and most experimental prion work on this subject have used transgenic animals that overexpress disease protein [65]. Transgenic mice, which have a foreign gene deliberately inserted in their genome, overexpress amyloid beta protein and are an incomplete and limited model for Alzheimer’s disease [66]. On the other hand tuberculosis and in particular its CWD forms were an integral part in conceptions regarding Alzheimer’s from its inception to the present. By 2013 Mawanda and Wallace pointed to two prime suspects for Alzheimer’s amyloid-beta deposition: “especially chronic infections like tuberculosis and leprosy” [67].
Recently, Alteri showed that during active infection, M. tuberculosis produces outer fringelike bacterial amyloid structures called “fimbriae” or “pilli” on its surface that also bind Congo red dye, a property associated with human amyloidosis [68]. Jordal reconfirmed that mycobacteria like TB produce a level of amyloid not previously described for any other bacteria. Not only was amyloid found present in tubercular mycobacterial biofilms, but in the actual cell envelops of the mycobacteria, as well as coating the spores they produce [69].
There is no known disease which better fits into what is occurring in Mad Cow, scrapie and the rest of the spongiform encephalopathies – including its human counterparts than the Mycobacteria tuberculosis complex and its blood-brain barrier penetrating, viral-like, cell-wall-deficient forms. It is for these reasons that future research needs to be aimed in this direction.
Acknowledgments Including Sources of Support
There were no sources of commercial funding or grants for this study.
Conflict of Interest
The authors have no conflict of interest, financial or otherwise with regard to this study. Procedures involving experimentation on animal subjects were done humanely and in accord with either the guide of the institution in which the experiments were done.
References
- Curto M, Reali C, Palmieri G, Scintu F, Schivo ML, et al. (2004) Inhibition of Cytokines Expression in Human Microglia Infected with Virulent and Nonvirulent Mycobacteria. NeurochemInternat 44: 381-392.
- Broxmeyer L, Sosnowska D, Miltner E, Chacón O, Wagner D, et al. (2002) Killing of Mycobacterium avium and Mycobacterium tuberculosis by a mycobacteriophage delivered by a nonvirulentmycobacterium: a model for phage therapy of intracellular bacterial pathogens. J Infect Dis 186: 1155-1160.
- Randall PJ, Hsu NJ, Lang D, Cooper S, Sebesho B, et al. (2014) Neurons are host cells for Mycobacterium tuberculosis. Infect Immun 82: 1880-1890.
- Manuelidis EE, Manuelidis L (1989) Suggested Links Between Different Types of Dementias: Creutzfeldt-Jakob disease, Alzheimer disease, and Retroviral CNS Infections. Alzheimer Dis AssocDisord 3: 100-109.
- Stockman S (1911) The habits of British ticks found on sheep and cattle. J Comp Pathol24: 229-37.
- Fischer O (1911) Thespongious loss of the bones, a special process of destruction of the cerebral cortex. Z gesNeurolPsychiat 7:1-33.
- Babes V, Levaditi C (1897) On the Actinomycotic Shape of the Tuberculosis Bacilli (Sur la FormeActinomycosique du Bacilli de la Tuberculosis). Arch of Med ExpetD’anat 9: 1041-1048.
- Dunkin GW (1936)Paratuberculosis of Cattle and Sheep. Section of Comparative Medicine. Proc R Soc Med 30:83-90
- Chauveau A: Transmission of virulent diseases by the ingestion of virulent principles in the digestive tract. Gaz de Paris: p 45.
- Klebs E (1870)On the history of tuberculosis.Virchows Arch F pathAnat :p 291
- Gerlach AC (1870)On the inoculability of tuberculosis and the perilla, and on the transferability of the latter by feeding. Virchows Arch F pathAnat 11: 297.
- Pfeiffer DU (1994)The role of a wildlife reservoir in the epidemiology of bovine tuberculosis. Massey University, Dept of Veterinary Clinical Sciences, New Zealand.
- Bourne J, Donnelly C (2001)An epidemiological investigation into bovine tuberculosis third report of the Independent Scientific Group on Cattle TB.
- Francis J (1947) Bovine Tuberculosis: Including a Contrast with Human Tuberculosis. Staples Press Limited, London: p 220.
- Taubes G (1986)The game of the name is fame. But is it science? Discover 7:28-52.
- Hadlow WJ, Prusiner SB, Kennedy RC, Race RE (1980) Brain tissue from persons dying of Creutzfeldt-Jacob disease causes scrapie-like encephalopathy in goats. Ann Neurol8: 628-631.
- Griffith JS (1967) Self-replication and scrapie. Nature215:1043-4.
- Prusiner SB (1995) The prion diseases. Sci Am 272: 48-51.
- Pethe K, Bifani P, Drobecq H, Sergheraert C, Debrie AS, et al. (2002) Mycobacterial heparin-binding hemagglutinin and laminin-binding protein share antigenic methyllysines that confer resistance to proteolysis. ProcNatlAcadSci USA 2002 99: 10759-10764.
- Tsubuki S, Takako Y (2003) Dutch, Flemish, Italian and Arctic mutations of App and resistance of Abeta to physiologically relevant proteolytic degradation. Lancet 361:1957-1958.
- Lasmézas CI, Deslys JP (1997) Transmission of the BSE agent to mice in the absence of detectable abnormal prion protein. Science 275: 402-405.
- Baker CA, Martin D, Manuelidis L (2002) Microglia from Creutzfeld–Jakob disease-infected brains are infectious and show specific mRNA activation profiles. J Virol76:10905-10915.
- Botsios S, Manuelidis L (2016) CJD and Scrapie Require Agent-Associated Nucleic Acids for Infection. J Cell Biochem 117:1947-1958.
- Abalos P, Retamal P (2004) Tuberculosis: a re-emerging zoonosis? Rev Sci Tech 23:583-94.
- O'Reilly LM, Daborn CJ (1995)The epidemiology of Mycobacterium bovis infections in animals and man: a review. Tuber Lung Dis 76:1-46.
- Grange JM (2001) Mycobacterium bovis infection in human beings. Tuberculosis 81:71-77
- Mattman LH (2001) Cell Wall Deficient Forms: Stealth Pathogens. CRC Press 3 (416).
- Xalabarder C (1958) Electron microscopy of tubercle bacilli. ExcerptaMedica. Sec XV Chest Dis 11: 467-473.
- Xalabarder C (1963) The Nature of So-Called Atypical Mycobacteria. Neumol Cir Torax24:259-74.
- Csillag A (1964) Themycococcus form of mycobacteria. Journ of Gen Microbio34: 341-352
- Shleeva MO, Salina EG, Kaprel'iants AS (2010)Dormant form of Mycobacterium tuberculosis. Mikrobiologiia 79:3-15
- Zhang Y, Yang Y, Woods A, Cotter RJ, Sun Z (2001) Resuscitation of dormant Mycobacterium tuberculosis by phospholipids or specific peptides. BiochemBiophys Res Commun284:542-547.
- Marcova N, Slavchev G, Michailova L (2012)Unique biological properties of Mycobacterium tuberculosis L-form variants: impact for survival under stress. Int. Microbiol15:61-68.
- Shleeva MO, Mukamolova GV, Telkov MV, Berezinskaia TL, Syroeshkin AV et al. (2003) Formation of nonculturable Mycobacterium tuberculosis and their regeneration. Mikrobiologiia 72:76-83.
- Prusiner SB (2014) Madness and Memory: The Discovery of Prions – A New Biologic Principle of Disease. Yale University Press.
- Hass G, Huntington R (1943) Amyloid. III The properties of amyloid deposits occurring in several species under diverse conditions. Arch Pathol35:226.
- Schwartz P (1972) Amyloid degeneration and tuberculosis in the aged. Gerontologia 18: 321-362.
- Delgado WA (1997) Amyloid deposits in labial salivary glands identified by electron microscopy. J Oral Pathol Med 26: 51-52.
- de Beer FC, Nel AE (1984) Serum amyloid A protein and C-reactive protein levels in pulmonary tuberculosis: relationship to amyloidosis. Thorax 39:196–200.
- Tomiyama T, Satoshi A (1994) Rifampicin prevents the aggregation and neurotoxicity of amyloid B protein in vitro. BiochemBiophys Res Commun204: 76-83.
- Chauhan A, Madiraju MV, Fol M, Lofton H, Maloney E, et al. (2006) Mycobacterium tuberculosis Cells Growing in Macrophages Are Filamentous and Deficient in FtsZ Rings. J. Bacteriol 188:1856-1865.
- Markova N, Michailova L, Kussovski V, Jourdanova M (2008) Formation of Persisting Cell Wall Deficient Forms of Mycobacterium bovis BCG during Interaction with Peritoneal Macrophages in Guinea Pigs. Electronic Journal of Biology 4: 1-10
- Thacore H, Willett HP (1966)The formation of spheroplasts of Mycobacterium tuberculosis in tissue culture cells. Am Rev Respir Dis 93:786-796.
- Lysenko AP, Vlasenko AP, Broxmeyer L (2014) Phenomenon of variability of mycobacteria and its use for detection of a tuberculosis infection.
- Lysenko AP, Vlasenko VV, Broxmeyer L, Lemish AP, Novik TP, et al. (2014) The tuberculin skin test: how safe is safe? The tuberculins contain unknown forms capable of reverting to cell-wall-deficient mycobacteria. Clinical and Experimental Medical Sciences 2: 55-73.
- Lysenko AP, Broxmeyer L, Vlasenko V, Krasochko PA, Lemish AP, et al. (2016) Further evidence for Cancer as Cell-wall-deficient Mycobacterial Disease. J MolPatholEpidemiol 1: 1-12.
- Lysenko AP, Vlasenko VV, Lemish AP (2014) Detection of mycobacteria in tis- sues by means of the differentiating immunoperoxidase staining. Tuberculos i boleznilegkhih. 10: 55-58.
- Harry EJ (2001) Bacterial cell division: regulating Z-ring formation. MolMicrobiol 40:795-803.
- Errington J, Daniel RA, Scheffers DJ (2003) Cytokinesis in bacteria. MicrobioMolBiol Rev 67:52-65.
- Xalabarder C (1970) L-forms of chronic mycobacteria and nephritis. PublInstAntituberc(Barcelona) Supple 7:7-83.
- Seidel B, Thomzig A, Buschmann A, Groschup MH, Peters R, et al. (2007) Scrapie agent (strain 263K) can transmit disease via the oral route after persistence in soil over years. PLoS One 2: e435.
- Ghodbane R, MbaMedie F, Lepidi H, Nappez C, Drancourt M (2014) Long-term survival of tuberculosis complex mycobacteria in soil. Microbiology160: 496-501.
- Insanov AB, Gadzhiev FS (1996) Comparative Analysis of the Results of Spinal Fluid Microbiological Study in Children and Adults Who Suffered from Tuberculous Meningitis. Probltuberk. 5: 25–28.
- Slavchev G, Michailova L, Markova N (2013) Stress-induced L-forms of M. bovis: challenge to survivability. New Microbiologica36:157-166.
- Calmette A, Valtis J, Lacomme A (1928)New experimental research on tuberculousultravirus. CR AcadSci 186: 1778-1781.
- Xiao X, Miravalle L, Yuan J, McGeehan J, Dong Z, et al. (2009) Failure to Detect the Presence of Prions in the Uterine and Gestational Tissues from a Gravida with Creutzfeldt - Jakob disease.Am J Pathol 174:1602-1608.
- Brieger EM (1949)The Host Parasite Relationship in Tuberculous Infection. Tubercle 30: 242-253.
- Brieger EM, Glauert AM (1952) A Phase-Contrast Study of Reproduction in Mycelial Strains of Avian Tubercle Bacilli. J GenMicrobiol 7: 287-294.
- Prusiner SB (2004) Development of the Prion Concept. Prion Biology and Diseases, Cold Spring Harbor Laboratory Press: pp89-141.
- Pattison IH (1988) Fifty years with scrapie: a personal reminiscence. Vet Rec 123:661-666.
- Pattison IH, Jones KM (1967)The possible nature of the transmissible agent of scrapie. Vet Rec 80:7.
- Gerston KF, Blumberg L, Tshabalala VA, Murray J (2004) Viability of mycobacteria in formalin-fixed lungs. Hum pathol 35: 571-575.
- Vinnie DS, Mary R (2002) Does formaldehyde kill Myco tuberculosis? Tech Bull Histopath 2: 37-38.
- Alzheimer A, Forstl H, Levy R (1991) On Certain Peculiar Diseases of Old Age. History of Psychiatry 2: 71-101.
- Goedert M (2015) Alzheimer’s and Parkinson’s diseases: The prion concept in relation to assembled Aß, tau and α-synuclein. Science 349:1255555.
- Schwab C, Hosokawa M, McGeer PL (2004)Transgenic mice overexpressing amyloid beta protein are an incomplete model of Alzheimer disease. ExpNeurol 188: 52-64
- Mawanda F, Wallace R (2013) Can infections cause Alzheimer's disease? Epidemiol Rev35:161-80.
- Alteri CJ, Xicahténcati-Cortes J, Hess S, Caballero-Olin G, GirónJA,et al. (2007) Mycobacterium Tuberculosis ProducesPili during Human Infection.ProcNatlAcadSci USA 104: 5145-5150.
- Jordal PB, Dueholm MS, Larsen P, Petersen SV, Enghild JJ, et al. (2009) Widespread abundance of functional bacterial amyloid in mycolata and other gram-positive bacteria. Appl Environ Microbiol 75: 4101-4110.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences